572bj Quantifying the Contractile Behavior of Individual Fibroblasts within Three-Dimensional Collagen Scaffolds: A Cellular Solids Approach
Cell-mediated contraction plays a critical role in many physiological and pathological processes, notably organized contraction during wound healing. Implantation of an appropriately formulated (i.e., mean pore size, chemical composition, degradation rate) three-dimensional scaffold into an in vivo wound site effectively blocks the majority of organized wound contraction and results in induced regeneration rather than scar formation [1]. Improved understanding of cell contraction within three-dimensional constructs therefore represents an important area of study in tissue engineering.
Studies of cell contraction within three-dimensional constructs typically calculate an average contractile force from the gross deformation of a macroscopic substrate by a large cell population [2]. In this study, cellular solids theory has been applied to conventional column buckling relationships in order to quantify the magnitude of individual cell contraction events within a three-dimensional, collagen-glycosaminoglycan (CG) scaffold. This new technique can be used for studying cell mechanics with a wide variety of porous scaffolds that resemble low-density, open-cell foams. It extends previous methods for analyzing cell buckling of two-dimensional substrates to three-dimensional constructs. The mean contractile force (Fc) generated by individual dermal fibroblasts within CG scaffolds was calculated to range between 11 and 41 nN (Fc = 26 ± 13 nN, Mean ± StDev), with an upper bound of cell contractility estimated at 450 nN.
Future iterations of this work will attempt to provide a more comprehensive treatment of cell contraction within 3D scaffolds and ECM analogs by integrating three-dimensional cell tracking using fluorescent technologies in order to incorporate the effect of loading eccentricity to this system and to better visualize cytoskeletal reorganization during contraction. Comparing the individual and populational cell contractile capacity within the CG scaffold may allow aspects of cell cooperativity to be probed and may suggest techniques for modifying the scaffold microstructure or mechanics to prevent organized scaffold (and therefore wound) contraction during in vivo wound healing and regeneration studies. The individual cell contraction technique adds a powerful new technique to the methods available to study cell contractility, particularly due to the ability to study individual cell contractility within a bioactive 3D scaffold with direct in vivo applications for induced tissue regeneration where understanding cell contraction is critical [1].
References:
1. Yannas IV. Tissue and Organ Regeneration in Adults. New York: Springer; 2001.
2. Freyman TM, Yannas IV, Yokoo R, Gibson LJ. Fibroblast contraction of a collagen-GAG matrix. Biomaterials 2001;22(21):2883-2891.
Keywords: fibroblast; contraction; force generation; biomaterial; cellular solid
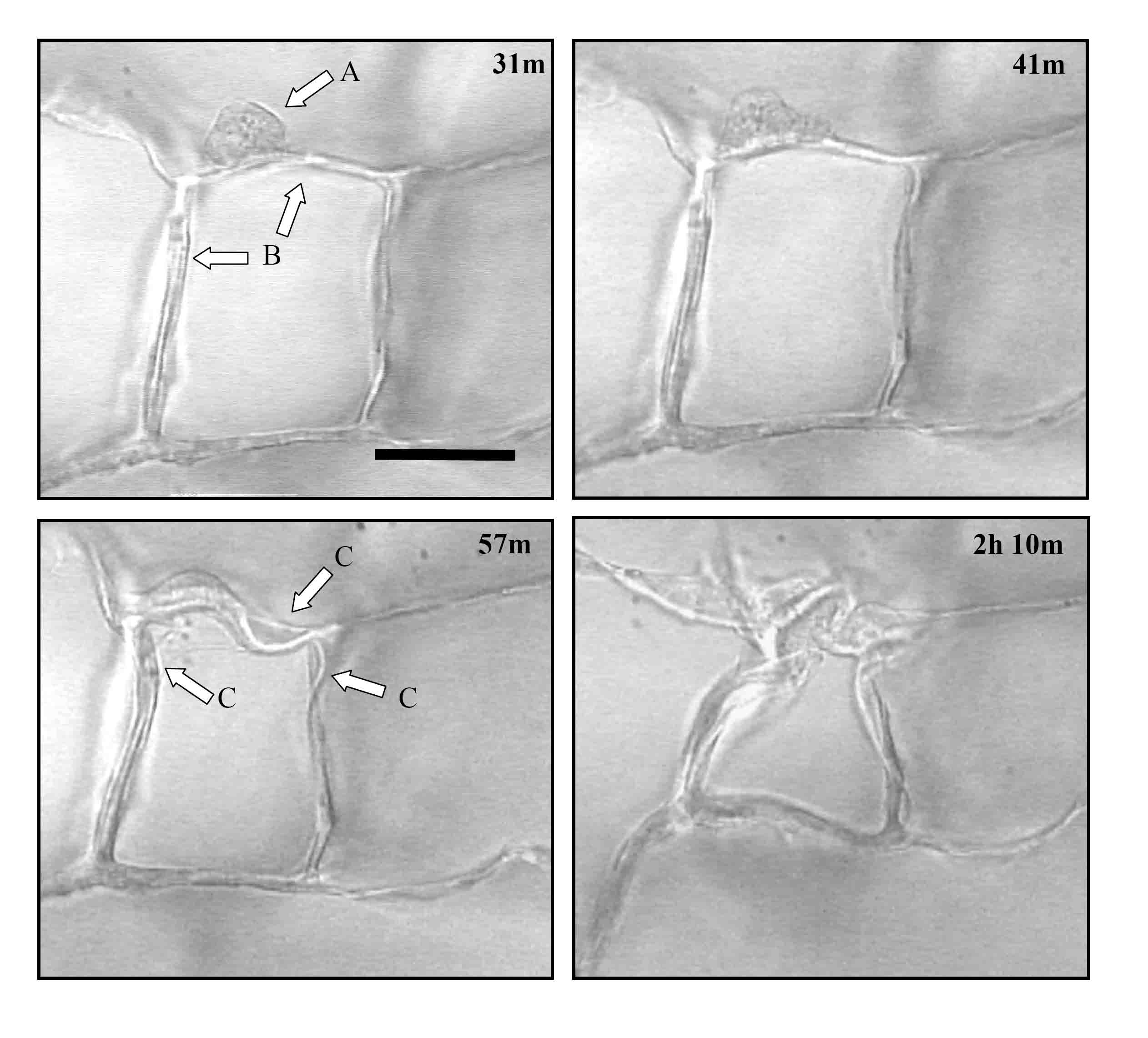
Figure 1. Time lapse images of an individual dermal fibroblast within the CG scaffold. The sequence of images shows a dermal fibroblast (arrow A) elongating and deforming the scaffold surrounding struts (arrows B). Several struts are deformed over time (arrows C). The number in the top right corner of each image indicates the time, in hours and minutes, after cell seeding. Scale bar: 50 µm. ADDIN EN.REFLIST